|
|
aHR0cDovL2ZyZWVzaGlwLmNvLmty
- 스마트 기기: no
- 모델 번호: 211212
- 백라이트 옵션: no
- 디스플레이 유형: ANALOG
- 브랜드 이름: MIRONG
- 근원: DE (원산지)
- 증명서: 세륨,DOT,EAC,EPA,FCC,GMP,ROHS,TGA,Ul,KC,pse,weee,NONE
- No.: 211212
- Color classification: 2mm does not invoice 2mm open ticket, 2mm open vote, 3mm no invoice, 3
- Decoration and construction contents: Installation engineering
- DIY Supplies: Electrical
gce Glassy carbon electrode Glass carbon, referred to as glassy carbon, is the slow heating of polyacrylonitrile resin or phenolic resin in an inert atmosphere to high temperature(reach1800C)The glassy carbon is treated as glassy amorphous carbon, an electronic conductor material suitable for use as an electrode, and also widely used in table tennis substrates. Glassy carbon electrode is the abbreviation of glass carbon electrode. The advantages of the glass carbon electrode are good conductivity, high chemical stability, small thermal expansion coefficient, firm texture, good air tightness and wide range of potential applications(From about-1~1VAnd saturated calomel electrode)The utility model can be made into an electrode shape of a cylinder, a disk, etc., and can be used as a substrate, and a merCury film, a glassy carbon electrode and a chemically modified electrode can be also made. It has been widely used in electrochemical experiments or electroanalytical chemistry. Electrode characteristics: The glassy carbon electrode is the working electrode of a wide range of uses, it is a kind of inert electrode good, has good conductivity, high hardness, high smoothness, high hydrogen overpotential, polarization range, chemical stability, can be directly used as inert electrode cathode and anodic stripping voltammetric determination of valence ions. Can also be used for chemical modified electrode. Electrode treatment and maintenance: The glassy carbon surface must be mirror and clean. Because the glass carbon surface vulnerable to some organic compounds pollution, which seriously affect the measurement (not a peak, a miscellaneous peak, not reproduce) before the measurement must be so clean, there are three main methods, chemical methodOneSoak and scrub with nitric acid.Twowith Ammonia ethanol or ethyl acetate1:1Soak scrub.ThreeCan also be washed with alcohol, and then6NHCLor4NHO3Soak. Electrochemical treatment: in+0.8V-(-One8V(0.5MKcLPH7exceptO2IUnder conditions (voltage), repetitive polarization (reset) within voltage range-Scan) (anode)-The cathode to the cathode) if it is seriously polluted and has a linen pit, scratches can be treated mechanically,MgO(powder200Put it on the wet flannelette and add a little water to polish it. Can also be Several methods are combined according to the electrode conditions. Should not soak the electrode for a long time, again strong acid, strong alkali and organic solvent. Because the glassy carbon electrode is an inert electrode, the scanning material used is scanning electrodes, such as mercury, copper, and gold. They are the mercury film, the copper film and the gold film electrode. For example: anodic stripping, mercury plating, and gold plating. There are two kinds of mercury in glassy carbon. The first is mercury plated with equal quantity, that is, a certain amount is added to the sample being analyzedHg+(generally used in mercuric oxide and mercuric nitrate) to the ion Electrolytic Enrichment point, while the formation of amalgam, scanning dissolution, in the positive potential, completely dissolved ions or mercury film to be measured, it can remove the mercury film electrode with filter paper wipe. When the second analysis is ensured, the electrode surface is still in the original state to ensure reproducibility, and the concentration of the common mercury ion is the concentration of the ion to be measured500-1000Second times for the mercury plated film,1.Mercury accumulated for a long time under the electrolytic potential of the ions to be measured. Such as the measurementcuIn-0.2VA mercury film is deposited over a long period of time to prevent electrodepositionCu, Pb, CD, ZnThe risk of this method is low efficiency and long time.TwoUnder the condition of equal mercury plating, after the electrolysis is made for a period of time, the scanning dissolves and dissolves at the end potential to dissolve the general time, thus dissolving the ions to be measured so as to be enriched-Dissolution, repeated several times, you can get a layer of mercury ions without measuring ions. as0.5MKCL+2 x 10-2MHg+ ten drops of saturated NaSO3, -1.4V to 5-10 minutes mixing concentration, stirring state in 15 seconds to scan to -0.1V, continue to stir the dissolution of 30 seconds in -0.1V, then in the reset state under stirring enrichment 5-10 minutes, 15 seconds stirring scan to -0.1V, then stir the dissolution of 30 seconds under -0.1V solution still take out electrode rinse the electrode rod to prevent coating break. Mercury plating should pay attention to the electrode under no bubble sorption, otherwise uneven plating coating mercury on the electrode without short-term soak in 1:1 nitric acid, then wash with plating, not long to be mercury washed, dried and preserved. The basic principle of glass carbon plating is the same as that of mercury plating.
|
|
|
|
|















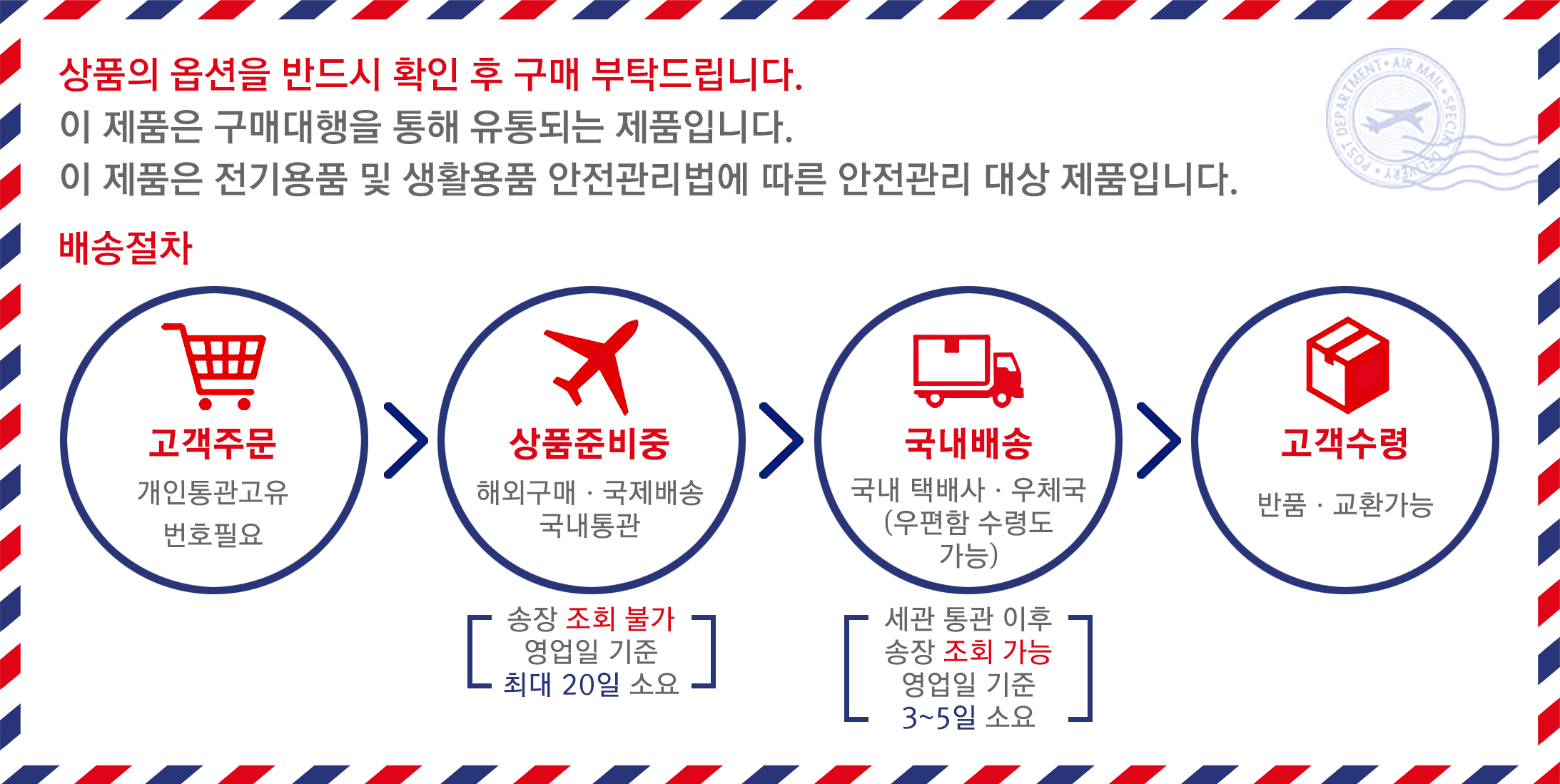
 배송기간
배송기간