|
|
  aHR0cDovL2ZyZWVzaGlwLmNvLmty aHR0cDovL2ZyZWVzaGlwLmNvLmty 
- 브랜드 이름: MIRONG
- 근원: CN (정품)
- 스마트 기기: YES
- 디스플레이 유형: ANALOG
- 백라이트 옵션: no
- 모델 번호: 3MM.4MM.5MM.
- 증명서: 세륨,DOT,EAC,EPA,FCC,GMP,ROHS,TGA,Ul,KC,pse,weee,NONE
Suggestion remarks: glass carbon electrode should be avoided as far as possible. The data is wrong, and the polishing is repaired.
Platinum carbon electrodeGlass carbon, abbreviated as glassy carbon, is a kind of electronic conductor material, which is slowly heated to high temperature (up to 1800 degrees C), such as polyacrylonitrile resin or phenolic resin in the inert atmosphere to high temperature (up to 1800 degrees C). It is also widely used in table tennis floor.The glassy carbon electrode is the abbreviation of the glass carbon electrode.The glass carbon electrode has the advantages of good conductivity, high chemical stability, small thermal expansion coefficient, hard texture, good air tightness, wide application range (about -1 to 1V, relative to saturated calomel electrode), which can be made into cylindrical and disk electrodes, and can be used as the matrix of mercury carbon glass carbon electrode and chemical modified electrode. It has been widely applied in electrochemical experiments or electroanalytical chemistry.Characteristics of electrode:The glassy carbon electrode is one of the widely used working electrodes. It is a good inert electrode with good conductivity, high hardness, high degree of finish, high hydrogen overpotential, wide polarization range, and chemical stability. It can be used as an inert electrode directly for anodic dissolution, volt ampere determination of cathode and variable valence, and can be used as a chemical repair. Ornament electrode.Electrode treatment and maintenance:The surface of glassy carbon must be guaranteed to be mirror and clean. Because the surface of glass carbon is easily polluted by some organic metal compounds, it seriously affects the measurement (no peak, out of peak, no recurrence), so cleaning treatment must be done before the measurement, the main methods are three kinds, chemical method 1. nitric acid soaking and scrubbing. 2.Ammonia water or ethyl acetate 1:1 immersion scrub. 3. can also be washed with alcohol and then soaked in 6NHCL or 4NHO3. Electrochemical treatment: in +0.8V- (-1.8V (0.5MKcLPH7 O2I) under the voltage range of repeated polarization (reset - scan) (anode to cathode to the cathode) if serious pollution and there is a pit, scratch can be mechanical treatment, MgO powder (more than 200 mesh) on the wet flannelette, plus a small amount of water polishing. Also canSeveral methods are used jointly according to the condition of the electrode. It is not suitable to immerse the electrode for a long time, strong acid, strong base and organic solvent.Because the glassy carbon electrode is an inert electrode, the scanning material is scanned electrode, such as mercury, copper, gold, mercury film, copper film and gold film electrode. For example: anodic dissolution of mercury plating and gold plating, there are two kinds of mercury plating mercury: the first kind of mercury plating is in the same position, that is to add a certain amount of Hg+ (general mercury oxide and mercuric nitrate) in the analyzed sample (general mercury oxide and mercury nitrate) at the point position which is enriched by electrolysis, and form amalgam at the same time. After scanning and dissolving out, it dissolves thoroughly to be measured at a more positive potential. The mercury can also be removed by using filter paper. It is guaranteed that the surface of the electrode is still original in the second analysis to ensure reproducibility, the concentration of mercury ions is 500-1000 times that of the measured ion concentration, the second kinds of mercury films are given, and 1. is electrodeposited for long time under the electrolysis potential of the pending ion. For example, Cu can produce mercury film in -0.2V long time electrowinning and prevent the danger of Cu, Pb, CD and Zn. This method is inefficient and long. 2. under the condition of mercury plating in the same position, after the electrodeposition was made for a period of time, the scanning was dissolved and the general time was dissolved under the terminated potential.Such as 0.5MKCL+2 x 10-2MHg+ more than ten drops of saturated NaSO3, stirring for 5-10 minutes in -1.4V, scanning to -0.1V in 15 seconds, stirring and dissolving for 30 seconds under -0.1V, then stirring and enriching for 5-10 minutes in the reset state, stirring in 15 seconds to -0.1V, then stirring in -0.1V for 30 seconds, and taking out the electrode after the solution is still. Use the electrode to remove the electrode after the solution is still Water flushed the electrode to prevent the coating from breaking. Mercury plating should not have bubble suction under the electrode, otherwise the mercury coating is not uniform and electroplating not, in the short term, the electrode can not be soaked in 1:1 nitric acid. The basic essentials of glassy carbon gold plating are the same as mercury plating.
|
|
|
|
|

















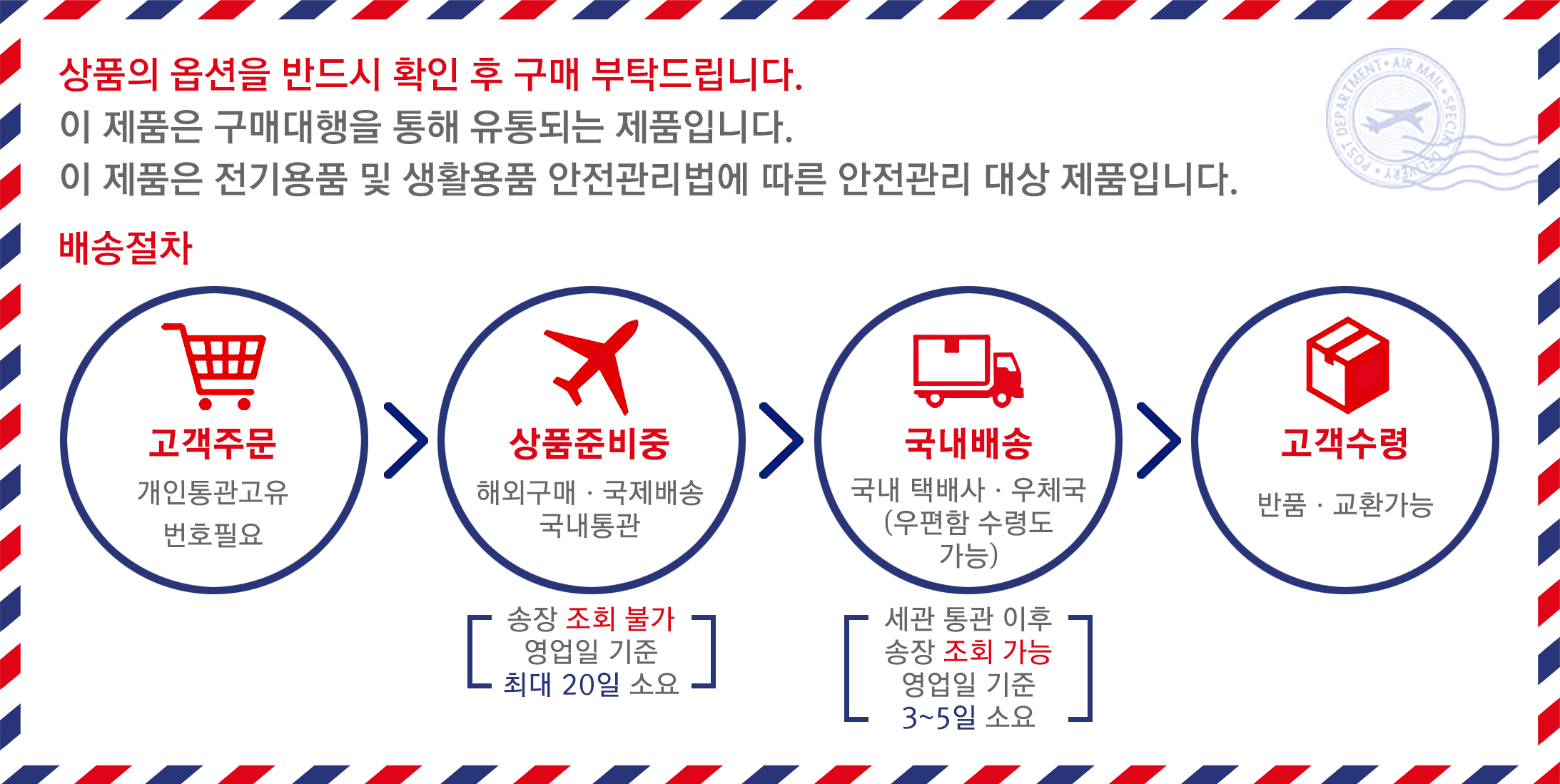
 배송기간
배송기간